 |
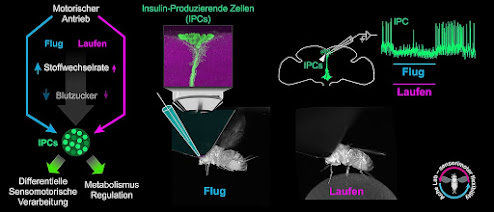
| The figure shows the relationships between movement and regulation of the insulin-producing cells in the fruit fly. Illustration Credit: Sander Liessem / University of Würzburg |
If a fruit fly starts to run or fly, its insulin-producing cells are immediately inhibited. This could explain why exercise promotes health.
Insulin is an essential hormone for humans and many other living things. Its most well-known task is to regulate sugar metabolism. How it does this job is well researched. Much less is known about how the activity of the insulin-producing cells and consequently the release of insulin is controlled.
A team from the Biozentrum of the Julius Maximilians University (JMU) Würzburg is now providing news on this question in the journal Current biology in front. The group of Dr. used as the object of investigation. Jan Ache the fruit fly Drosophila melanogaster. Interestingly, this fly also releases insulin after a meal - but the hormone does not come from the pancreas like in humans, but from nerve cells in the brain.
Electrophysiological measurements for active flies
The JMU group found that physical activity of the fly has a strong effect on its insulin-producing cells. For the first time, the researchers have the activity of these cells electrophysiologically during running or flying Drosophila measured.
Result if Drosophila when it starts to run or fly, their insulin-producing cells are quickly inhibited. If the fly ends its movements, the activity of the cells immediately increases again and at times even shoots beyond the normal value.
"We believe that the low activity of the insulin-producing cells during the movement contributes to the provision of sugar to meet the increased energy requirements," says Dr. Sander Liessem, first author of the publication. “And we suspect that the increased activity after the movement has ended helps to replenish the fly's energy stores, for example in the muscles."
Blood sugar is irrelevant to the regulation
The JMU team was also able to demonstrate that the very rapid behavior-dependent inhibition of insulin-producing cells is actively controlled by nerve tracts. "It is largely independent of changes in the sugar concentration in the blood of the animals," explains co-author Dr. Martina hero.
It makes a lot of sense for the organism to anticipate an increasing energy requirement in this way in order to prevent extreme fluctuations in the blood sugar level.
Insulin has hardly changed in evolution
Allow the results to draw conclusions about humans? Probably yes.
"Although insulin is released into fruit flies via cells other than humans, the insulin molecule and its function have hardly changed in the course of evolution," says Jan Ache. And have been with you in the past 20 years Drosophila As a model organism, many basic questions have already been answered, which could also contribute to a better understanding of metabolic defects in humans and associated clinical pictures such as diabetes or obesity.
Less insulin means longevity
"An exciting point is that reduced insulin activity contributes to healthy aging and longevity," says Sander Liessem. This has already been demonstrated in flies, mice, humans and other species. The same applies to an active lifestyle. “Our work shows a possible connection here, how physical activity via neural signaling pathways could have a positive effect on insulin regulation."
Further steps in research
Next, Jan Ache's team wants to investigate which messenger substances and circuits of the nervous system in the fly are responsible for changing the activity of the insulin-producing cells. This should be challenging: Several messenger substances are usually involved in neuromodulation processes, and the individual substances can have opposite or complementary effects in combination.
The group now analyzes the diverse ways in which insulin-producing cells receive input from outside. She also examines other factors that could affect the activity of these cells, such as the age of the flies or their nutritional status.
"In parallel, we are researching neuronal control of running behavior," explains Jan Ache. The long-term goal of his group is to bring these two research questions together: how does the brain control running and other behaviors, and how is it ensured that the energy balance is regulated accordingly?
Published in journal: Current biology
Source/Credit: University of Würzburg | Von Robert Emmerich
Reference Number: bio010423_01
