 |
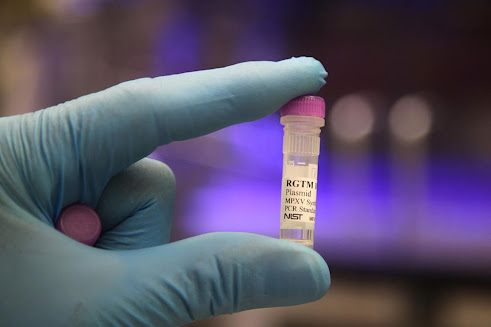
| A vial of the positive control material from NIST that can be used to help ensure the accuracy of tests for monkeypox. Credit: R. Press/NIST |
In an effort to help speed the expansion of monkeypox testing in the U.S., the National Institute of Standards and Technology (NIST) has produced a material that can help ensure the accuracy of tests for the disease. NIST is making the material, which contains gene fragments from the virus that causes the disease but is noninfectious and safe to handle, freely available for use by test manufacturers and testing laboratories.
Monkeypox is spread by close contact and can cause fever, flu-like symptoms and skin lesions. More than 3,500 cases of monkeypox have been confirmed in the United States since the outbreak began in late May, and the World Health Organization has declared monkeypox to be a global health emergency.
Testing is necessary to identify the extent of an outbreak and contain it, and to properly care for people who have caught the disease and those who may have been exposed. The monkeypox test, like the most sensitive test for COVID-19, uses a technique called polymerase chain reaction, or PCR, to detect genetic sequences from the virus that causes the disease.
Because the material from NIST contains those genetic sequences, laboratories can use it as a positive control — that is, a sample that should cause a positive result if their test is working properly. As the U.S. Centers for Disease Control and Prevention (CDC) works to expand the nation’s testing capacity, the material from NIST will fill a growing need.

























.jpg)
