 |
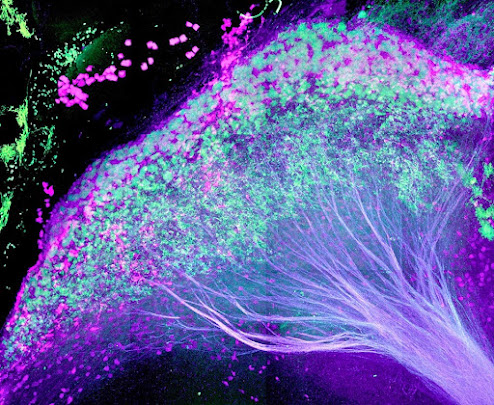
| The lateral habenula in the mouse brain, with axons streaming down to dopaminergic and serotonergic centers. Credit: Warden Lab |
A tiny but important area in the middle of the brain acts as a switch that determines when an animal is willing to work for a reward and when it stops working, according to a study published Aug. 31 in the journal Current Biology.
“The study changes how we think about this particular brain region,” said senior author Melissa Warden, assistant professor and Miriam M. Salpeter Fellow in the Department of Neurobiology and Behavior, which is shared between the College of Arts and Sciences and the College of Agriculture and Life Sciences.
“It has implications for psychiatric disorders, particularly depression and anxiety,” Warden said.
The paper, “Tonic Activity in Lateral Habenula Neurons Acts as a Neutral Valence Brake on Reward-Seeking Behavior,” illuminates the role of the lateral habenula, a small structure on top of the thalamus, which funnels higher-level information from the front and center of the brain to areas that produce neurotransmitters such as serotonin and dopamine.
The lateral habenula’s exact role has been unclear until now. The new study shows that when neurons in this brain area turn off, an animal will work for rewards; when those neurons fire, the animal becomes disengaged and stops working. Experiments revealed that the lateral habenula turns on specifically when an animal has had enough of a reward and is satisfied, or when it finds its work no longer yields a reward.














.jpg)

