Three-dimensional semiconductor particles have an edge, too – many of them – given their geometrically varied surfaces. Cornell researchers have discovered that the junctures at these facet edges have 2D properties, which can be leveraged for photoelectrochemical processes – in which light is used to drive chemical reactions – that can boost solar energy conversion technologies.
This research, led by Peng Chen, the Peter J.W. Debye Professor of Chemistry in the College of Arts and Sciences, could also benefit renewable energy technologies that reduce carbon dioxide, convert nitrogen into ammonia, and produce hydrogen peroxide.
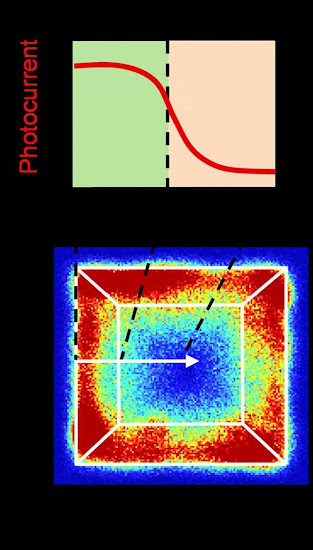
A high-resolution map of a photocatalyst particle shows the transition zones of reactivity and the corresponding spatial variation of photoelectrochemical performance across the inter-facet edge.
The group’s paper, “Inter-Facet Junction Effects on Particulate Photoelectrodes,” published in Nature Materials. The paper’s lead author is postdoctoral researcher Xianwen Mao.
For their study, the researchers focused on the semiconductor bismuth vanadate, particles of which can absorb light and then use that energy to oxidize water molecules – a clean way of generating hydrogen as well as oxygen.
The semiconductor particles themselves are anisotropically-shaped; that is, they have 3D surfaces, full of facets angled toward each other and meeting at edges on the particle surface. However, not all facets are equal. They can have different structures that, in turn, result in different energy levels and electronic properties.















.png)





.jpg)