 |
| Magma ocean and first rocky crust on the Moon Credit: NASA/Goddard Space Flight Center |
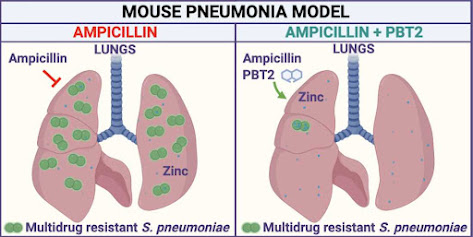
Over fifty years ago, Apollo 11 astronauts collected samples from the lunar Highlands. These large, pale regions of the Moon – visible to the naked eye – are made up of relatively light rocks called anorthosites. Anorthosites formed early in the history of the Moon, between 4.3 and 4.5 billion years ago.
Similar anorthosites, formed through the crystallization of magma, can be found in fossilized magma chambers on Earth. Producing the large volumes of anorthosite found on the Moon, however, would have required a huge global magma ocean.
"Cooling of the early magma ocean drove such vigorous convection that crystals remained suspended as a slurry, like the crystals in a slushy machine."Jerome Neufeld
Scientists believe that the Moon formed when two protoplanets, or embryonic worlds, collided. The larger of these two protoplanets became the Earth, and the smaller became the Moon. One of the outcomes of this collision was that the Moon was very hot – so hot that its entire mantle was molten magma, or a magma ocean.














.jpg)